Tissue‑repair peptides: mechanisms, assays, and practical guidance for research labs
In a university tendon-repair lab I visited, two graduate students ran parallel cohorts: full‑thickness skin wounds treated with a peptide gel and identically wounded controls. They photographed wounds every 48 hours, measured tensile strength at day 21, and ran qPCR for VEGF and collagen I. The peptide arm closed faster and showed different collagen organization on histology. That protocol—simple, repeatable, oriented around clear endpoints—is what this article is about.
What we mean by “tissue‑repair peptides” “Tissue‑repair peptides” is an umbrella term for short amino‑acid chains that modify cellular behaviours relevant to wound healing: cell migration, angiogenesis, inflammation resolution, and extracellular matrix remodelling. Many are native fragments or analogues of endogenous proteins (for example, a gastric‑juice fragment or a small copper‑binding tripeptide). Others are synthetic mimetics designed to concentrate a functional motif. Researchers use them to probe biological mechanisms, to create biomaterials with biologically active coatings, or to test combination strategies with growth factors or scaffolds. The goals vary: accelerate epithelial closure, improve tensile properties of healed tissue, or reduce scarring by altering collagen deposition. All experimental claims should be limited to the model system studied; none of the discussion here recommends or implies human use.
Core mechanisms observed across models Although each peptide has its own profile, several recurring mechanistic themes show up in cell culture and animal work:
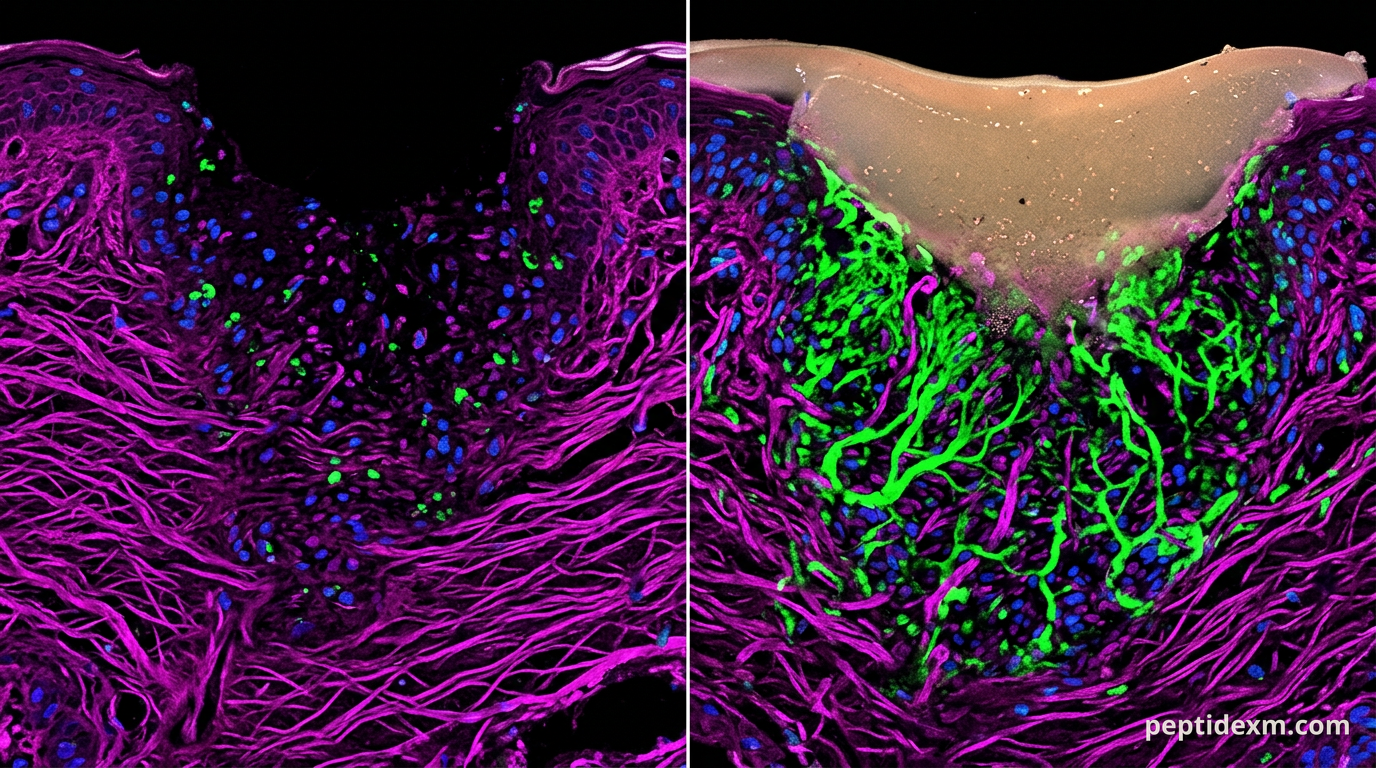
Angiogenesis modulation: increased endothelial migration, tube formation in vitro, and higher CD31/VEGF expression in tissues. Cell migration and proliferation: fibroblasts, keratinocytes, and endothelial cells often exhibit faster scratch‑assay closure or increased BrdU/EdU incorporation. ECM remodelling: altered ratios of collagen I to collagen III, changes in MMP (matrix metalloproteinase) activity, and different collagen fibril alignment on polarized light microscopy. Inflammation modulation: reductions in acute proinflammatory cytokines (IL‑6, TNF‑α) and sometimes earlier transition to a resolution phenotype with higher IL‑10 expression.
Molecular targets and common markers When planning assays, choose markers that map to the mechanism you care about. Common readouts include:
Angiogenesis: VEGF (mRNA/protein), CD31 immunostaining, microvessel density counts. Migration/proliferation: scratch/wound closure assays, transwell migration, Ki‑67 or BrdU labelling. ECM: collagen I/III ratio by qPCR or western blot, hydroxyproline content, MMP‑2/MMP‑9 zymography. Inflammation: IL‑1β, IL‑6, TNF‑α, IL‑10 by ELISA or multiplex assays; macrophage phenotype markers (CD68, CD163).
Spotlight: BPC‑157 — practical notes for bench researchers BPC‑157 is a 15‑amino‑acid peptide derived from a gastric‑juice protein. In vitro and multiple rodent models it has been associated with faster epithelial and tendon healing, altered angiogenic signalling, and modulation of inflammatory markers. Those findings are model‑specific and depend on formulation, timing, and assay choice. Typical experimental endpoints where BPC‑157 has been reported to show effects include wound closure rates in full‑thickness skin models, tensile testing in tendon injury models, and histological measures of collagen deposition and alignment. If you plan to include it in a biomaterial coating or local gel, stability in the formulation and release kinetics are critical variables to test.

Spotlight: Thymosin beta‑4 (TB‑500) — what to expect in assays Thymosin beta‑4 (Tβ4) is a 43‑amino‑acid actin‑binding protein; TB‑500 generally refers to a synthetic peptide derived from Tβ4’s active sequence. In multiple tissue models TB‑500 has been associated with enhanced cell migration and increased angiogenesis markers, and with changes in scar formation in animal studies. Standard readouts that have been used with TB‑500 include epithelial wound closure, endothelial tube formation in Matrigel, and collagen organisation on Masson’s trichrome staining. As with most peptides, delivery route matters: topical application to a wound bed behaves differently than a bolus applied systemically in an acute injury model.
Other peptides commonly used in tissue‑repair experiments A few additional sequences appear frequently in the literature and are worth keeping in the toolkit.
GHK‑Cu — a gly‑his‑lys tripeptide that forms a complex with copper: studied for skin remodelling, collagen synthesis, and anti‑oxidant responses. Useful when the endpoint is dermal ECM organisation or keratinocyte behaviour. KPV — a Lys‑Pro‑Val tripeptide derived from alpha‑melanocyte stimulating hormone; reported to affect inflammation and epithelial barrier function in mucosal models. Thymosin alpha‑1 — an immunomodulatory peptide used in immune‑system focused studies that sometimes pairs with repair protocols to probe host response to injury. PEG‑MGF and IGF‑1 variants — more focussed on muscle repair and hypertrophy, but occasionally used in composite repair approaches where muscle or tendon interface is assessed.
Designing experiments: models, endpoints, and controls Good experimental design separates signal from noise. Peptide experiments are sensitive to timing, delivery, and matrix effects. Choose a model that matches the biological question. Full‑thickness excisional skin wounds (rodents) are straightforward for measuring epithelial closure and collagen remodelling. Tendon transection models give tensile readouts. For angiogenesis, dorsal skinfold chambers or Matrigel plug assays provide quantifiable vessel growth.
Practical protocol considerations Here are concrete points that often determine whether results are interpretable.
Controls: include vehicle controls that match any carrier or gel. If using a scaffold, test scaffold alone, scaffold+peptide, and peptide alone where feasible. Randomisation and blinding: randomise animals to groups and blind outcome assessors (histology scoring, image analysis) to reduce bias. Sample size: power calculations for continuous outcomes (wound area, tensile strength) prevent underpowered studies; typical small‑animal studies often use n=6–12 per group depending on expected variance. Time points: collect acute (24–72 hour) and later remodelling time points (14–28 days) to capture both inflammatory changes and ECM maturation. Multiparameter endpoints: combine macroscopic (closure rate), biomechanical (tensile testing), molecular (qPCR/ELISA), and histological (collagen staining, vessel counts) readouts for a convincing story.
Assays and quantitative readouts that work well Choose assays appropriate to the mechanism you hypothesise. A single positive result is not usually definitive; concordant results across orthogonal assays are stronger.
Wound area measurement: calibrated digital photography and automated image analysis. Report % area reduction over baseline. Tensile testing: peak force to failure and stiffness (report units: N and N/mm or MPa where cross‑section is measured). Histology: Masson’s trichrome or Picrosirius red with polarized light for collagen orientation and maturity. Immunohistochemistry: CD31 (endothelial), alpha‑SMA (myofibroblasts), F4/80 or CD68 (macrophages). Molecular assays: qPCR for VEGF, TGF‑β1, collagen I/III, MMPs; zymography for MMP activity; multiplex cytokine panels for inflammatory profiling. Functional in vitro assays: scratch assays, transwell migration, and Matrigel tube formation for endothelial cells.
Handling, formulation, and stability — practical bench tips Short peptides can be deceptively easy to mishandle. Pay attention to solvent, adsorption to plastics, and freeze–thaw cycles.
Solvent choice: many peptides reconstitute in sterile water, phosphate buffer, or low‑percent DMSO for hydrophobic sequences. Avoid strong acids or bases unless validated for your peptide. Adsorption: peptides can stick to polypropylene tubes. Use low‑binding tubes for dilute solutions and consider adding a carrier protein (e.g., 0.1% BSA) for in vitro assays if compatible with endpoints. Storage: aliquot stock solutions to avoid repeated freeze–thaw. Short peptides often store well at −20 °C as lyophilised powder; for long‑term storage, −80 °C is safer for aqueous stocks. Formulation: for local delivery, test release kinetics from hydrogels or scaffolds using in vitro release assays (syringe extraction + HPLC or mass spec quantification). Sterility: if applying to open wounds in animal models, ensure sterility by sterile filtration when feasible; note that filtration can retain aggregates depending on molecular size.
Safety, reporting, and regulatory notes for research use All peptide work should be performed under appropriate animal‑use and biosafety approvals. Report methodology thoroughly: peptide sequence, supplier lot number, purity, method of reconstitution, vehicle composition, and storage conditions. These details materially affect reproducibility. Safety for lab personnel matters. Use appropriate PPE when handling powders and solutions; avoid aerosolisation. If an adverse event occurs in an animal model, document it fully and consult your institutional veterinarian or safety officer.
Combining peptides with other interventions: synergy and pitfalls Researchers often test peptides alongside growth factors, stem cells, or biomaterials. That can magnify effects, but it complicates interpretation. When combined interventions change multiple pathways, isolating mechanism requires careful controls and, ideally, factorial study designs. Beware of overlapping signalling: a peptide that increases VEGF and a growth factor that does the same may produce an additive angiogenic signal. That could be desirable for a tissue repair model, but it also increases the chance of off‑target effects in a complex system. In vitro dose–response and sequential dosing studies help clarify interactions before moving to in vivo combination experiments.
Common pitfalls and troubleshooting Negative or inconsistent results are often methodological rather than biological. Typical issues include poor peptide stability in the vehicle, inadequate concentration at the target site, or insensitive endpoints.
If you see no effect in vitro, verify peptide identity and purity (HPLC or mass spec) and confirm activity in a simple positive control assay (e.g., endothelial tube formation for angiogenic peptides). If in vivo results are noisy, increase sample size, add earlier time points, or tighten inclusion criteria for animals (age, sex, weight) to reduce biological variance. When histology is equivocal, use quantitative image analysis rather than subjective scoring. Thresholding and blinded quantification improve reproducibility.
Back in that tendon lab, the protocol that mattered most was not the peptide name but the rigor around endpoints: consistent wounds, blinded scoring, and multiple orthogonal assays. Tissue‑repair peptides can be powerful experimental tools when handled carefully. The data they produce are only as useful as the experimental design that generated them.